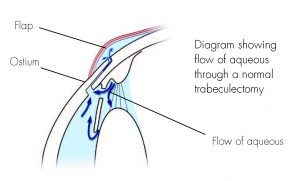
Image of trabeculectomy
INDICATION
Mitosol® (mitomycin for solution) 0.2 mg/vial Kit for Ophthalmic Use is an antimetabolite indicated as an adjunct to ab externo glaucoma surgery.
Dosage & Administration
Mitosol® is intended for topical application to the surgical site of glaucoma filtration surgery and must be reconstituted prior to application. Sponges provided within the Mitosol® kit should be fully saturated with the entire reconstituted contents in a manner prescribed in the Instructions For Use. The sponge(s) should be applied to the treatment area for two minutes. Reconstituted Mitosol® should be used within one hour of reconstitution.
US Patents #7,806,265, #8,186,511, #D685,962, #D685,963, #9,205,075, #9,539,241 and #9,649,428; other international patents issued and pending.
Product Information
Please click here for full Product Information for Mitosol®
IMPORTANT SAFETY INFORMATION
Contraindications
Mitosol® is contraindicated in patients that have demonstrated a hypersensitivity to mitomycin in the past.
Warnings & Precautions
Cell Death: Mitomycin is cytotoxic. Use of mitomycin in concentrations higher than 0.2 mg/mL or use for longer than 2 minutes may lead to unintended corneal and/or sclera damage including thinning or perforation. Direct contact with the corneal endothelium will result in cell death. Hypotony: The use of mitomycin has been associated with an increased instance of post-operative hypotony. Cataract Development: Use in phakic patients has been correlated to a higher instance of lenticular change and cataract formation. Embryo-Fetal Toxicity: Can cause fetal harm. Advise of potential risk to a fetus. Verify pregnancy status in females of reproductive potential prior to use.
Adverse Events & Reactions
The most frequent adverse reactions to Mitosol® occur locally and include hypotony, hypotony maculopathy, blebitis, endophthalmitis, vascular reactions, corneal reactions, and cataract.
You are encouraged to report all side effects to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088. You may also call Glaukos at 1-888-404-1644.